Ok. Probably a really dumb question but..... So I've heard about people using Temp Control which my mod can do with the right coils obviously. But when its set to say 600F or even 200F is that the heat of the coil or the actual vapor produced. Because wouldn't even 200F vapor cause steam burns. I mean... Try holding your hand over a pot of boiling 212F water.... Anyone know the science behind this? Sorry if this sounds dumb.
Become a Patron!
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Regarding TC Mode Safety
- Thread starter ember2018
- Start date
-
- Tags
- burns safety
That is the internal heat of wire in the coil calculated some way I can't explain. If you check the temperature of the vapor coming off a coil you would be surprised at how cool it actually is when the MOD is set for say 460°F in TC Mode.
Temperature Control (TC) is the mode through which the box/mod regulates the intensity in order to keep the coil at a temperature that is defined by the user. According to the manufacturers, maintaining the temperature constant is the guaranty of more safety but this argument is sometimes misused since, for example, no indication is given, neither on devices nor on juice bottles, regarding the temperature to use.
Basic knowledge on material science
A coil is most of the time a resistive wire that is folded in a spring-like cylinder with non-touching circular turns. The wire’s resistivity (in Ω.m-1) defines how strongly a given wire, generally an alloy, opposes to the flow of electrons, or electric current. Heat is produced when electrical energy is dissipated into the wire when electron are scattered by the atoms present in the alloy, the so-called Joule effect
The joule effect is a physical law that describes the relationship between the heat generated and the current flowing through a conductor.
The temperature reached by the coil is directly proportional to the square of the current intensity (U2) and inversely proportional to the value of the resistance (R).
Application: For a given resistance coil (value in Ω), a higher temperature is reached when increasing the intensity (voltage). But if you keep the same voltage, a lower temperature will be obtained with a higher resistance value.
">Joule effect.
When you purchase a coil from a vendor, it comes with a label showing its resistance, a value in Ohm (Ω).
The resistance of wires changes with temperature
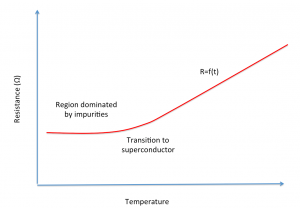
Relationship between resistance (R) and temperature (T) for a resistance coil
The resistance of an alloy is stable near room temperature and generally increases with temperature as the material becomes a superconductor. Then, and only after this transition, its resistance can be predicted from its temperature.
It is also possible to use the revert relationship to reach a setpoint temperature. And this is exactly the principle that uses your favorite mod when switching to temperature control.
The temperature control
As a general rule, the resistance (R) of a coil increases with temperature (T). In the temperature control mode, the chipset of the box measures on a very fast rate the resistance when incrementing intensity levels. The chipset basically increases the intensity of the current up to reaching a certain value (R) of the resistance. This setpoint value (R) corresponds to the desired temperature (T).
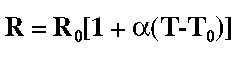
Relationship between the Resistance (R) and the temperature (T) (see the text for more information).
However, to obtain this temperature (T) with more or less of accuracy, the procedure strongly depends on the calibration parameters.
In the calibration procedure (generally an operation that is made manually and that is more or less described in the user manual), the nominal value of the resistance (R0) is measured by the device at room temperature (T0) and the temperature coefficient of resistance (α) is selected. For the sake of simplicity, manufacturers offer the possibility of select α from the type of resistive wire used: Nickel (Ni), Titanium (Ti) or Stainless steel (SS).
In some more expert modes, α can be entered as a numerical value. This allows using types of alloys that are not common or to adapt to new resistive coils to come on the market without changing one’s box.
When is it recommended to use the TC mode?
Ni, Ti and SS coils are compliant with temperature control but not Kanthal. The reason why Kanthal does not allow temperature control is because this FeCrAl alloy has much more impurities than any others and does not offer superconductivity properties (a linear relationship of resistance with temperature). In fact, the electrons are so scattered by the impurities that they heat the alloy while the resistance is kept constant. Special grades of Kanthal can be used as high as 1,425°C in the industry.
Is the temperature control precise and accurate?
No. But it is better than nothing.
The parameter α is fitted from measurement data in a defined experimental setting that does not necessarily meet the conditions that are used with an e-cigarette. Hence, α is dependent on T0. Since the box is not equipped to measure this temperature, α is set as a constant by the manufacturer. Hence, if you calibrate your box at 17°C or 27°C, the accuracy of the T:R relationship will vary. Remember that the linear approximation is only an approximation and is different for different reference temperatures T0.
Such warnings should call users to use caution when selecting their working temperature and to keep a bit of freedom with what they choose as their higher limit.
What temperature to select?
The lowest as possible… Right, but depending on your device and the juice you vape, a minimum of heat is needed to produce vapor. In mixes, different liquids are present with different properties and boiling points.
Water boils at 100°C, Propylene Glycol (PG
Propylene glycol
">PG) is vaporized at 188°C (or 371°F), Vegetal Glycerin (VG
Vegetable Glyceryn
Vegetable Glycerin is a colorless, odorless, and tasteless viscous liquid that is used in pharmaceutical formulations. VG is exclusively derived from plants (soybeans or palm), since some can also be of synthetic origine or come from animal fat. Glycerol is an additive (E422) used in the food industry and a thickening agent or a preservative. Its sweet taste makes it a sugar substitute present in low-fat products.
">VG) at 290°C (or 554°F) and ethanol at 78°C (or 173°F).
Other constituents are also present whose concentration and composition are also very variable from one juice to another.
The most common is nicotine whose concentration cannot exceed 2% in Europe, but with values up to 4.8% in certain juices like those used in the Vype ePen, for example. Nicotine’s boiling point is reached at 247°C (or 477 °F).
Flavorings can reach up to 20% in volume of the final blend. Diacetyl
Diacetyl or butane-2,3-dione ((CH3CO)2) is an organic compound that is a by-product of fermentation.
Diacetyl is naturally present in alcoholic beverages and is used as a buttery flavor in the food industry. This flavor is also used in the elaboration of receipes of e-liquids.
Despite innocuousness when ingested, diacetyl has proven harmfulness upon inhalation. Some employees of manufactures using artificial butter flavoring have been diagnosed with bronchiolitis obliterans, a rare and serious disease of the lungs associated to this compound.
Together with acetylpropionyl, diacetyl is of legal use as a flavoring substance in the European food industry but their use is controversial in e-liquids for their potential harmfulness (Farsalinos et al., 2014).
">Diacetyl, for example, that gives a buttery taste, boils at only 88°C (or 190°F), for acetoin giving the same flavor, it is higher (148°C or 298°F). For the flavor of banana, isoamyl acetate is vaporized at 142°C (or 288°F). Benzaldehyde (cherry taste) boils at 178°C (or 352°F) and cinnamaldehyde (cinnamon taste) at 248°C (478°F). Fruity flavors (ethyl propionate) boils at 99°C (or 210°F), grape (Methyl anthranilate) at 256°C (or 493°F), orange (limonene) at 176°C (or 349°F), Pinnaple (allyl hexanoate) at 190°C (or 374°F), cotton candy (ethyl maltol) at 161°C (or 322°F), menthol at 212°C (or 414°F) and vanilla (vanilline) at 295°C (or 563°F).
Basic knowledge on material science
A coil is most of the time a resistive wire that is folded in a spring-like cylinder with non-touching circular turns. The wire’s resistivity (in Ω.m-1) defines how strongly a given wire, generally an alloy, opposes to the flow of electrons, or electric current. Heat is produced when electrical energy is dissipated into the wire when electron are scattered by the atoms present in the alloy, the so-called Joule effect
The joule effect is a physical law that describes the relationship between the heat generated and the current flowing through a conductor.
The temperature reached by the coil is directly proportional to the square of the current intensity (U2) and inversely proportional to the value of the resistance (R).
Application: For a given resistance coil (value in Ω), a higher temperature is reached when increasing the intensity (voltage). But if you keep the same voltage, a lower temperature will be obtained with a higher resistance value.
">Joule effect.
When you purchase a coil from a vendor, it comes with a label showing its resistance, a value in Ohm (Ω).
The resistance of wires changes with temperature

Relationship between resistance (R) and temperature (T) for a resistance coil
The resistance of an alloy is stable near room temperature and generally increases with temperature as the material becomes a superconductor. Then, and only after this transition, its resistance can be predicted from its temperature.
It is also possible to use the revert relationship to reach a setpoint temperature. And this is exactly the principle that uses your favorite mod when switching to temperature control.
The temperature control
As a general rule, the resistance (R) of a coil increases with temperature (T). In the temperature control mode, the chipset of the box measures on a very fast rate the resistance when incrementing intensity levels. The chipset basically increases the intensity of the current up to reaching a certain value (R) of the resistance. This setpoint value (R) corresponds to the desired temperature (T).
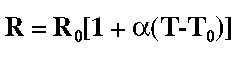
Relationship between the Resistance (R) and the temperature (T) (see the text for more information).
However, to obtain this temperature (T) with more or less of accuracy, the procedure strongly depends on the calibration parameters.
In the calibration procedure (generally an operation that is made manually and that is more or less described in the user manual), the nominal value of the resistance (R0) is measured by the device at room temperature (T0) and the temperature coefficient of resistance (α) is selected. For the sake of simplicity, manufacturers offer the possibility of select α from the type of resistive wire used: Nickel (Ni), Titanium (Ti) or Stainless steel (SS).
In some more expert modes, α can be entered as a numerical value. This allows using types of alloys that are not common or to adapt to new resistive coils to come on the market without changing one’s box.
When is it recommended to use the TC mode?
Ni, Ti and SS coils are compliant with temperature control but not Kanthal. The reason why Kanthal does not allow temperature control is because this FeCrAl alloy has much more impurities than any others and does not offer superconductivity properties (a linear relationship of resistance with temperature). In fact, the electrons are so scattered by the impurities that they heat the alloy while the resistance is kept constant. Special grades of Kanthal can be used as high as 1,425°C in the industry.
Is the temperature control precise and accurate?
No. But it is better than nothing.
The parameter α is fitted from measurement data in a defined experimental setting that does not necessarily meet the conditions that are used with an e-cigarette. Hence, α is dependent on T0. Since the box is not equipped to measure this temperature, α is set as a constant by the manufacturer. Hence, if you calibrate your box at 17°C or 27°C, the accuracy of the T:R relationship will vary. Remember that the linear approximation is only an approximation and is different for different reference temperatures T0.
Such warnings should call users to use caution when selecting their working temperature and to keep a bit of freedom with what they choose as their higher limit.
What temperature to select?
The lowest as possible… Right, but depending on your device and the juice you vape, a minimum of heat is needed to produce vapor. In mixes, different liquids are present with different properties and boiling points.
Water boils at 100°C, Propylene Glycol (PG
Propylene glycol
">PG) is vaporized at 188°C (or 371°F), Vegetal Glycerin (VG
Vegetable Glyceryn
Vegetable Glycerin is a colorless, odorless, and tasteless viscous liquid that is used in pharmaceutical formulations. VG is exclusively derived from plants (soybeans or palm), since some can also be of synthetic origine or come from animal fat. Glycerol is an additive (E422) used in the food industry and a thickening agent or a preservative. Its sweet taste makes it a sugar substitute present in low-fat products.
">VG) at 290°C (or 554°F) and ethanol at 78°C (or 173°F).
Other constituents are also present whose concentration and composition are also very variable from one juice to another.
The most common is nicotine whose concentration cannot exceed 2% in Europe, but with values up to 4.8% in certain juices like those used in the Vype ePen, for example. Nicotine’s boiling point is reached at 247°C (or 477 °F).
Flavorings can reach up to 20% in volume of the final blend. Diacetyl
Diacetyl or butane-2,3-dione ((CH3CO)2) is an organic compound that is a by-product of fermentation.
Diacetyl is naturally present in alcoholic beverages and is used as a buttery flavor in the food industry. This flavor is also used in the elaboration of receipes of e-liquids.
Despite innocuousness when ingested, diacetyl has proven harmfulness upon inhalation. Some employees of manufactures using artificial butter flavoring have been diagnosed with bronchiolitis obliterans, a rare and serious disease of the lungs associated to this compound.
Together with acetylpropionyl, diacetyl is of legal use as a flavoring substance in the European food industry but their use is controversial in e-liquids for their potential harmfulness (Farsalinos et al., 2014).
">Diacetyl, for example, that gives a buttery taste, boils at only 88°C (or 190°F), for acetoin giving the same flavor, it is higher (148°C or 298°F). For the flavor of banana, isoamyl acetate is vaporized at 142°C (or 288°F). Benzaldehyde (cherry taste) boils at 178°C (or 352°F) and cinnamaldehyde (cinnamon taste) at 248°C (478°F). Fruity flavors (ethyl propionate) boils at 99°C (or 210°F), grape (Methyl anthranilate) at 256°C (or 493°F), orange (limonene) at 176°C (or 349°F), Pinnaple (allyl hexanoate) at 190°C (or 374°F), cotton candy (ethyl maltol) at 161°C (or 322°F), menthol at 212°C (or 414°F) and vanilla (vanilline) at 295°C (or 563°F).
Manufacturers are often tempted to add exhausters in order to better develop certain flavors. They use sweeteners like sugars among which the most common is the saccharose that decomposes at 186°C (or 367°F). Glutamic acid is another popular exhauster (also called glutamate) that decomposes at 199°C (390°F). The reaction between an amine group of the glutamic amino-acid and a reducing sugar leads to the Maillard reaction
The Maillard reaction takes its name from the French physician and chemist, Camille Louis Maillard, who first described, in 1921, the reactions that involve amino acids and sugars. It occurs in a wide range of situation and, for example, is observed when roasting a piece of meat on the barbecue at high temperature or at lower temperature in ageing of wine.
Browning and the Maillard reaction
Browning and the Maillard reaction co-occur in samples, the browning being rather attributed to the effect of pH on the sugars [1]. The Maillard reaction rather takes place in alkaline solutions.
In e-liquids left steeping
The Maillard reaction is observed when aliments become brownish when cooking and render the typical odors of cooking like roasted meat, brown onions, bread, caramel...
The Maillard reaction and browning likely happen also in e-liquids left steeping for a long time. From transparent, a liquid turns golden then brown even in the absence of light and at room temperature. Depending on the flavors added, the browning may or may not occur. The presence of nicotine (that becomes brown for other reasons) tends to accelerate the overall browning of the blend.
From a chemical point of view, a carbonyl group of the sugar reacts with a nucleophilic amino group of the amino acid and forms a mixture of uncharacterized molecules responsible for a range of odors and flavors spanning from smoothly roasted to completely carbonized food.
By-products of the Maillard reaction
By-products of the Maillard reaction include glycosylamines, ketosamines, acetyl-, diacetyl and a wide diversity of aldehydes. Generally occurring around 140°C to 165°C (285°F to 330°F), other products are formed at higher temperature, among which acrylamide, a carcinogen that is also present in cigarette smoke.
[1] Schroeder, L. J., Iacobellis, M., & Smith, A. H. (1955). The influence of water and pH on the reaction between amino compounds and carbohydrates. Journal of Biological Chemistry, 212(2), 973-984.">Maillard reaction that produces a wide variety of flavors when products are heat.
Hence, the optimum temperature is difficult to assess due to the variety of reactions and the different compounds that are met. Based on readings on various forums, users generally opt for a temperature value around 250°C for puffs shorter than 5 seconds. At a lower value, the coil does not heat homogeneously andThey evoke the “cooking” of their juice when gunk build up on the wick, which is generally associated to a degraded flavor. Sometimes, flavor degradation is extreme and leads to a repulsion that expert users associate to the production of acrolein, a molecule that it is preferable to avoid.
Build your own
It is possible to build your own coils by purchasing a roll of resistive wire and to fashion it yourself. Take a small cylinder (a precision screwdriver is a good starting point) and turn your wire around it. Generally 6 turns are good. Some mounting decks, e.g. the Velocity
A mounting plate Velocity-type is inspired from the Velocity dripper. It is made of + and - connectors with two wholes each and allows dual coil mountings.
">velocity-type, are specially dedicated to home-made coils and allow vapers to set up their coil in the atomizer
The atomizer or "atty" is the part of the e-cigarette that produces the vapor. It is shaped as a cylindric tank that receives the e-liquid in which soaks the wick(s).
The e-liquid climbs by capillarity to the coil, a resistive wire that is heated when powered.
">atomizer.
When building your own coil, the number of turns imports since it defines the length of resistive wire and defines the value of its resistance. It is also important to avoid contact between the turns because the electrons that are in motion when the electric circuit is closed always choose the most direct way to go from one electrode to the other.
To be fully operational, insert the wick into the coils and cut the tips at the right size so that they dip into the liquid. Before using it for the first time, let your atomizer rest for a while. It requires a dozen of minutes before the juice climbs by capillarity and soaks the wick.
Google it to find many different explanations.
The Maillard reaction takes its name from the French physician and chemist, Camille Louis Maillard, who first described, in 1921, the reactions that involve amino acids and sugars. It occurs in a wide range of situation and, for example, is observed when roasting a piece of meat on the barbecue at high temperature or at lower temperature in ageing of wine.
Browning and the Maillard reaction
Browning and the Maillard reaction co-occur in samples, the browning being rather attributed to the effect of pH on the sugars [1]. The Maillard reaction rather takes place in alkaline solutions.
In e-liquids left steeping
The Maillard reaction is observed when aliments become brownish when cooking and render the typical odors of cooking like roasted meat, brown onions, bread, caramel...
The Maillard reaction and browning likely happen also in e-liquids left steeping for a long time. From transparent, a liquid turns golden then brown even in the absence of light and at room temperature. Depending on the flavors added, the browning may or may not occur. The presence of nicotine (that becomes brown for other reasons) tends to accelerate the overall browning of the blend.
From a chemical point of view, a carbonyl group of the sugar reacts with a nucleophilic amino group of the amino acid and forms a mixture of uncharacterized molecules responsible for a range of odors and flavors spanning from smoothly roasted to completely carbonized food.
By-products of the Maillard reaction
By-products of the Maillard reaction include glycosylamines, ketosamines, acetyl-, diacetyl and a wide diversity of aldehydes. Generally occurring around 140°C to 165°C (285°F to 330°F), other products are formed at higher temperature, among which acrylamide, a carcinogen that is also present in cigarette smoke.
[1] Schroeder, L. J., Iacobellis, M., & Smith, A. H. (1955). The influence of water and pH on the reaction between amino compounds and carbohydrates. Journal of Biological Chemistry, 212(2), 973-984.">Maillard reaction that produces a wide variety of flavors when products are heat.
Hence, the optimum temperature is difficult to assess due to the variety of reactions and the different compounds that are met. Based on readings on various forums, users generally opt for a temperature value around 250°C for puffs shorter than 5 seconds. At a lower value, the coil does not heat homogeneously andThey evoke the “cooking” of their juice when gunk build up on the wick, which is generally associated to a degraded flavor. Sometimes, flavor degradation is extreme and leads to a repulsion that expert users associate to the production of acrolein, a molecule that it is preferable to avoid.
Build your own
It is possible to build your own coils by purchasing a roll of resistive wire and to fashion it yourself. Take a small cylinder (a precision screwdriver is a good starting point) and turn your wire around it. Generally 6 turns are good. Some mounting decks, e.g. the Velocity
A mounting plate Velocity-type is inspired from the Velocity dripper. It is made of + and - connectors with two wholes each and allows dual coil mountings.
">velocity-type, are specially dedicated to home-made coils and allow vapers to set up their coil in the atomizer
The atomizer or "atty" is the part of the e-cigarette that produces the vapor. It is shaped as a cylindric tank that receives the e-liquid in which soaks the wick(s).
The e-liquid climbs by capillarity to the coil, a resistive wire that is heated when powered.
">atomizer.
When building your own coil, the number of turns imports since it defines the length of resistive wire and defines the value of its resistance. It is also important to avoid contact between the turns because the electrons that are in motion when the electric circuit is closed always choose the most direct way to go from one electrode to the other.
To be fully operational, insert the wick into the coils and cut the tips at the right size so that they dip into the liquid. Before using it for the first time, let your atomizer rest for a while. It requires a dozen of minutes before the juice climbs by capillarity and soaks the wick.
Google it to find many different explanations.
I'll definately re read that multiple times to get a better grasp but thanks for all the info. Definitely learning something new today. So my only question left is at the hottest what is the actual temp of the vapor as it exits the tip and enters the mouth? Being as saliva protects the mouth more than if it was dry skin it will probably be a tad hotter than I'm expecting. (People always think im crazy putting candles out with my fingertips but i always wet them first. Heh heh.)Manufacturers are often tempted to add exhausters in order to better develop certain flavors. They use sweeteners like sugars among which the most common is the saccharose that decomposes at 186°C (or 367°F). Glutamic acid is another popular exhauster (also called glutamate) that decomposes at 199°C (390°F). The reaction between an amine group of the glutamic amino-acid and a reducing sugar leads to the Maillard reaction
The Maillard reaction takes its name from the French physician and chemist, Camille Louis Maillard, who first described, in 1921, the reactions that involve amino acids and sugars. It occurs in a wide range of situation and, for example, is observed when roasting a piece of meat on the barbecue at high temperature or at lower temperature in ageing of wine.
Browning and the Maillard reaction
Browning and the Maillard reaction co-occur in samples, the browning being rather attributed to the effect of pH on the sugars [1]. The Maillard reaction rather takes place in alkaline solutions.
In e-liquids left steeping
The Maillard reaction is observed when aliments become brownish when cooking and render the typical odors of cooking like roasted meat, brown onions, bread, caramel...
The Maillard reaction and browning likely happen also in e-liquids left steeping for a long time. From transparent, a liquid turns golden then brown even in the absence of light and at room temperature. Depending on the flavors added, the browning may or may not occur. The presence of nicotine (that becomes brown for other reasons) tends to accelerate the overall browning of the blend.
From a chemical point of view, a carbonyl group of the sugar reacts with a nucleophilic amino group of the amino acid and forms a mixture of uncharacterized molecules responsible for a range of odors and flavors spanning from smoothly roasted to completely carbonized food.
By-products of the Maillard reaction
By-products of the Maillard reaction include glycosylamines, ketosamines, acetyl-, diacetyl and a wide diversity of aldehydes. Generally occurring around 140°C to 165°C (285°F to 330°F), other products are formed at higher temperature, among which acrylamide, a carcinogen that is also present in cigarette smoke.
[1] Schroeder, L. J., Iacobellis, M., & Smith, A. H. (1955). The influence of water and pH on the reaction between amino compounds and carbohydrates. Journal of Biological Chemistry, 212(2), 973-984.">Maillard reaction that produces a wide variety of flavors when products are heat.
Hence, the optimum temperature is difficult to assess due to the variety of reactions and the different compounds that are met. Based on readings on various forums, users generally opt for a temperature value around 250°C for puffs shorter than 5 seconds. At a lower value, the coil does not heat homogeneously andThey evoke the “cooking” of their juice when gunk build up on the wick, which is generally associated to a degraded flavor. Sometimes, flavor degradation is extreme and leads to a repulsion that expert users associate to the production of acrolein, a molecule that it is preferable to avoid.
Build your own
It is possible to build your own coils by purchasing a roll of resistive wire and to fashion it yourself. Take a small cylinder (a precision screwdriver is a good starting point) and turn your wire around it. Generally 6 turns are good. Some mounting decks, e.g. the Velocity
A mounting plate Velocity-type is inspired from the Velocity dripper. It is made of + and - connectors with two wholes each and allows dual coil mountings.
">velocity-type, are specially dedicated to home-made coils and allow vapers to set up their coil in the atomizer
The atomizer or "atty" is the part of the e-cigarette that produces the vapor. It is shaped as a cylindric tank that receives the e-liquid in which soaks the wick(s).
The e-liquid climbs by capillarity to the coil, a resistive wire that is heated when powered.
">atomizer.
When building your own coil, the number of turns imports since it defines the length of resistive wire and defines the value of its resistance. It is also important to avoid contact between the turns because the electrons that are in motion when the electric circuit is closed always choose the most direct way to go from one electrode to the other.
To be fully operational, insert the wick into the coils and cut the tips at the right size so that they dip into the liquid. Before using it for the first time, let your atomizer rest for a while. It requires a dozen of minutes before the juice climbs by capillarity and soaks the wick.
Google it to find many different explanations.
I once checked the temperature of the vapor coming off a coil set to 460°F and it was hundreds of degrees lower since evaporation starts taking place immediately. Now depending on Wattage and Temperature you have the MOD set for will definitely translate to the temperature of the vapor. Way too many variables for anyone to say what temperature your vapor will be.
Remember there is NO Need to be cranking up the wattage to get a good vape using TC. I like to set it where I can take long pulls and not hit Temperature Protect to quickly. Good Luck
Remember there is NO Need to be cranking up the wattage to get a good vape using TC. I like to set it where I can take long pulls and not hit Temperature Protect to quickly. Good Luck
R
Thanks! Was just curious as a ballpark figure of what that temp is actually.I once checked the temperature of the vapor coming off a coil set to 460°F and it was hundreds of degrees lower since evaporation starts taking place immediately. Now depending on Wattage and Temperature you have the MOD set for will definitely translate to the temperature of the vapor. Way too many variables for anyone to say what temperature your vapor will be.
Remember there is NO Need to be cranking up the wattage to get a good vape using TC. I like to set it where I can take long pulls and not hit Temperature Protect to quickly. Good Luck
Thanks! Was just curious as a ballpark figure of what that temp is actually.
When I first started with using TC I was pointed in many wrong directions by well meaning people. One thing you MUST always get right is having the Mod set for the right type of wire you are using for TC. Some exotic coils with mixed metals will deliver some seriously nasty vapes when not properly configured for the correct wire.
I
I got ya. Thanks for that. What I was curious to is the temp of the vapor coming out of any vaping device which im sure varies widely based on 100 different factors of course. I guess i could ask the temperature of the smoke you inhale from a cigarette but google says that its only about 10 degrees hotter than body temp. So 98.6 plus 10 is 108.6 well cool enough not to burn your mouth. Im going to assume the vapor is roughly the same. Sorry this just peaked my curiosity today. But now I got lots of good science stuff to look over and study that I never really even thought to explore before. I'm always trying to learn new things so I appreciate the info! I've learned about general a/c repair car repair general electrical stuff vaping types of batteries mainly nimh and li ion. And a whole mess of other probably useless information this past 6 months heh. But it keeps my mind going and helps keep my mind off my anxieties so I guess it's all positive! Maybe TC will be my next learning project.When I first started with using TC I was pointed in many wrong directions by well meaning people. One thing you MUST always get right is having the Mod set for the right type of wire you are using for TC. Some exotic coils with mixed metals will deliver some seriously nasty vapes when not properly configured for the correct wire.
