| |||||||||
| |||||||||
Become a Patron!
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Jimi's Daily Health Articles
- Thread starter Jimi
- Start date
|
|
Watch Del Bigtree break down this historic win here:

To share this legal update, please use this link: https://www.icandecide.org/ican_press/breaking-news-ican-obtains-cdc-v-safe-data/

To share this legal update, please use this link: https://www.icandecide.org/ican_press/breaking-news-ican-obtains-cdc-v-safe-data/
| Hello Jimi , We have a special announcement down below. It's time sensitive also. We’re nearing the end of our AMAZING 9-Episode docuseries, BRAVE: Live Couragously, Heal Miraculously… And the feedback we’ve received from thousands of viewers has been incredible… We want to say a heartfelt “THANK YOU” … With your support, we’ve been able to overcome Big Tech’s relentless censorship, break through the corporate media’s lies, and bring you the TRUTH… “This protocol … can be and has been demonstrated to be an extremely effective monotherapy for even advanced C0V!D, much less early C0V!D. And unfortunately … like most of these treatments, they've been ignored by mainstream medicine. I think that's one of the great frauds of medicine… they just … keep down natural therapies.” Dr. Thomas E. Levy Dr. Levy is one of the top medical experts you’ll hear from in today’s BRAND NEW Episode 8 BRAVE BREAKTHROUGHS: Latest Treatments to Overcome Vaxx Injuries |
‘Fully vaccinated’ people at 44% higher risk of COVID infection than unvaccinated: UK study
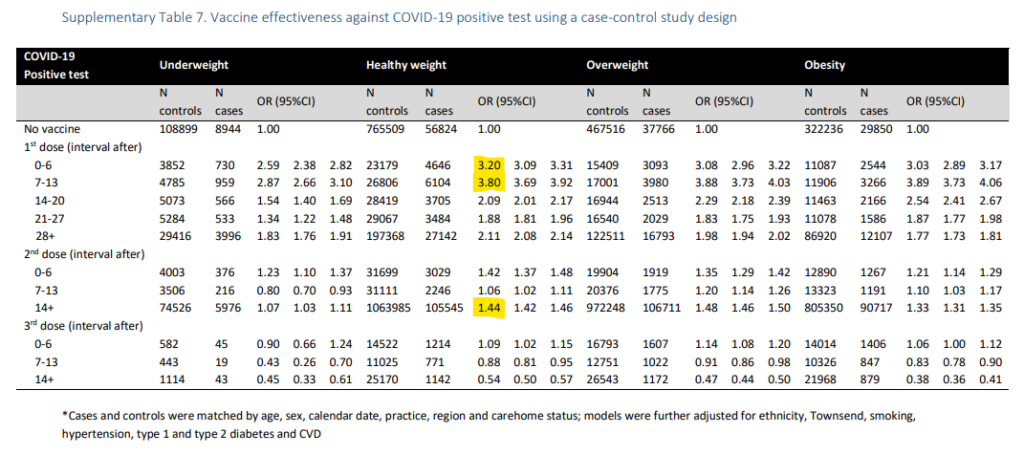
‘Fully vaccinated’ people at 44% higher risk of COVID infection than unvaccinated: UK study - LifeSite
Oxford University researchers found that those who had taken the anti-COVID shots stood at substantially higher risk of infection with the coronavirus compared to those who had did not receive any COVID jab.
‘Brilliant’ new documentary exposes how Big Pharma and gov’t teamed up to push the COVID vaccines

‘Brilliant’ new documentary exposes how Big Pharma and gov't teamed up to push the COVID vaccines - LifeSite
'Safe and Effective: A Second Opinion' chronicles the 'perfect storm” of safety trial problems, regulatory lapses, and government propaganda that led to mass 'vaccination' with a shot that has devastated lives.
| ||
Tuesday
October-4-2022'Maybe I'm not good enough', 'No one will ever love me', or 'I'm not worthy.'
Romantic rejection and ghosting can cause such negative thoughts and painful emotions to surface.
Dealing with rejection is hard. Give yourself plenty of time to feel all your feelings.
Once you let them run their course, you can focus on getting yourself to a better place and reflecting on your experience.
Remember, your worth doesn't come from outside approval. Avoid putting that person on a pedestal.
Just because the other person didn't want to pursue anything romantically with you, doesn't mean you're unworthy.
Separate your self-worth from the relationship. Use this experience to build resilience and understand what you want in a future relationship.
Best Lab Tests for Thyroid Problems

Best Lab Tests for Thyroid Problems — Bridgit Danner, Functional Health Coach & Detox Expert
Many people question whether their thyroid is working correctly due to their chronic fatigue, weight gain or foggy thinking. In this article, we’ll explore when and how to order a test for thyroid problems effectively so that you can address thyroid problems & symptoms.
|
Hi Jimi, Dr. Ritamarie here…
My clinically proven framework for assessing and resolving chronic fatigue promises to change how you look at fatigue.
I’d love to share it with you, but first I want to make sure you’re registered for my workshop…
Sign up now—it’s free to join
If you've been waiting a long time to find effective strategies to finally become the practitioner your patients and clients have been looking for, now is your chance to make that happen!
If you’re like me, you don’t want to waste your time on things that don’t work.
So, I’ve put together this workshop to teach you how to get to the root causes and what non-invasive, natural approaches you can use to empower your patients and clients to heal!
Chronic fatigue is a serious and debilitating condition that affects millions of people around the world.
It’s critical that you possess the skills and knowledge to properly identify & address its underlying causes—and to be able to provide real, lasting results!
The information, tips, and strategies you’ll discover in this training might just be the missing pieces in your practice…
Click here to see the details
I’ll see you later.
My clinically proven framework for assessing and resolving chronic fatigue promises to change how you look at fatigue.
I’d love to share it with you, but first I want to make sure you’re registered for my workshop…
Sign up now—it’s free to join
If you've been waiting a long time to find effective strategies to finally become the practitioner your patients and clients have been looking for, now is your chance to make that happen!
If you’re like me, you don’t want to waste your time on things that don’t work.
So, I’ve put together this workshop to teach you how to get to the root causes and what non-invasive, natural approaches you can use to empower your patients and clients to heal!
Chronic fatigue is a serious and debilitating condition that affects millions of people around the world.
It’s critical that you possess the skills and knowledge to properly identify & address its underlying causes—and to be able to provide real, lasting results!
The information, tips, and strategies you’ll discover in this training might just be the missing pieces in your practice…
Click here to see the details
I’ll see you later.
Deep Sleep: What Is It and How to Measure and Optimize It

Deep Sleep: What Is It and How to Measure and Optimize It
Deep sleep is a critical sleep stage for brain detoxification and healing. In this article. you will learn how to optimize your deep sleep.
Cholesterol: What is It and What are Healthy Levels?

Cholesterol: What is It and What are Healthy Levels?
Cholesterol is one of the most important molecules in your body and it is key to know its many functions and how to determine healthy levels.
Top 7 Herbs To Support Healthy Blood Sugar Levels

Top 7 Herbs To Support Healthy Blood Sugar Levels
In this article, I will discuss the importance of healthy blood sugar levels, and the top herbs that can help to support healthy blood sugar.
They say, "The devil is in the details." However, I like to turn that around and say, "The angel is in the details," because that's where you'll find the most helpful insights.
On that note, Jim, because you certainly want to be "in-the-know" on the best steps you can take to boost your daily energy, your mood, and your immunity, I'm certain you're going to appreciate...
The 5 top simple and natural ways you can increase your energy, improve your mood, and rejuvenate your immunity in Dr. Perlmutter and Dr. Bland's new free guide for you right here.
On that note, Jim, because you certainly want to be "in-the-know" on the best steps you can take to boost your daily energy, your mood, and your immunity, I'm certain you're going to appreciate...
The 5 top simple and natural ways you can increase your energy, improve your mood, and rejuvenate your immunity in Dr. Perlmutter and Dr. Bland's new free guide for you right here.
|
5 Food-Medicines That Could Quite Possibly Save Your Life
5 Food-Medicines That Could Quite Possibly Save Your Life
Some of the most powerful medicines on the planet are masquerading around as foods and spices. While they do not lend themselves to being patented, nor will multi-billion dollar human clinical trials ever be funded to prove them efficacious, they have been used since time immemorial to both...
|

|
|
|
|
How to Stop Vitamin C from Forming Oxalate
In a recent email I discussed potential issues that can arise when taking high doses of synthetic vitamin C (ascorbic acid or ascorbate), including the formation of oxalate, which can cause pain, mitochondrial function, oxidative stress, inflammation, and kidney stones.Oxalate can be formed from vitamin C after it gets oxidized to the ascrobyl radical and is further metabolized ultimately to oxalate.
This does not happen if oxidized vitamin C gets recycled back to ascorbic acid.
NADH and NADPH (the body's main reducing agent / electron donor) and glutathione recycle vitamin C.
NADPH is produced from glucose via the pentose phosphate pathway, and the efficiency of this process depends on various factors, including ...
- Metabolic issues that affect the availability of glucose
- Genetics affecting various enzymatic steps in the pentose phosphate pathway that produces NADPH
- Nutrients that act as cofactors in this pathway, including riboflavin, niacin, selenium, calcium, magnesium
- Diabetes, thyroid disorders, and adrenal disorders
- Oxidative stress
- Deficiencies in nutrients involved in energy metabolism (e.g. B vitamins, electrolytes, iron, copper, sulfur, and magnesium)
- Genetic variants affecting energy metabolism (too many to name here)
How well thioredoxin reductase uses NADPH to recycle vitamin C can be affected by the genes that code for this enzyme (TXNRD1 and TXNRD2 genes).
If vitamin C does not get recycled by NADPH, then it falls to glutathione to do it via glutathione-utilizing enzymes like glutathione S-transferases, which means that variants in certain GST genes can affect how well glutathione can recycle vitamin C.
If glutathione does not recycle vitamin C, then oxidized vitamin C can be irreversibly converted via several steps to oxalate.
However, oxidized vitamin C can also be excreted in the urine as a metabolite that has not yet converted all the way to oxalate.
So in general, oxalate formation from vitamin C occurs when NADPH / thioredoxin reductase and glutathione / glutathione S-transferase both fail to recycle it, but not all oxidized vitamin C necessarily converts to oxalate before getting excreted.
Oxalate can accumulate in the body if the body's pathways for converting oxalate to other substances (e.g. the amino acid glycine) are not working well.
The conversion of oxalate to other substances involves genes like LDHA, LDHB, LDHC, LDHD, AGXT, GRHPR and cofactors like vitamin B6.
In general, maintaining good NADPH and glutathione status seems important when engaging in high-dose vitamin C therapy, except when a prooxidant effect is desired (e.g. in cancer therapy) or when treating a dangerous condition like snake venom poisoning or polio infection that requires rapid treatment that is best accomplished immediately and without delay, using high-dose vitamin C.
In such cases, the risk of high-dose vitamin C therapy increasing oxidative stress or causing oxalate formation pale in comparison to the risk of death or permanent dysfigurement.
In general, oxalate formation from high-dose vitamin C therapy can most likely be prevented by ....
- Maintaining good levels of NADPH and glutathione
- Ensuring the availability of glucose (e.g. supporting insulin sensitivity, ensuring thryoid and adrenal function, and possibly not relying on a ketogenic diet if the body is struggling to produce enough glucose)
- Supplying he nutrients that are required in the relevant metabolic pathways
- Potentially also emplying extra measures to compensate for any genetic vulnerabilities in relevant metabolic pathways
There are reasons to employ caution, reasons to not worry excessively about it, and also steps that can be taken to minimize problems with high-dose vitamin C therapy.
Keep in mind that infection, stress, and toxins (including bacterial endotoxin), appear to cause vitamin C to leave the adrenal glands (the richest reservoir of vitamin C in the body) to go to the liver (to deal with toxins) and other parts of the body (e.g. the thymus gland to support immune function).
Depletion of vitamin C from the adrenal glands due to chronic infection or toxicity may be a contributor to fatigue caused by reduced adrenal function.
Ultimately, vitamin C is a powerful neutralizer of many types of infection and toxicity and also needs to be replenished in the adrenal glands.
Whether vitamin C is taken only in food-based forms or also in high-dose synthetic forms, vitamin C is very important in both the prevention of and recovery from infection and toxicity.
When it comes to assessing your risk for potential side effects from vitamin C supplementation, it can be helpful to look at variants in genes like G6PD, TXNRD1, TXNRD2, GST, LDHA, LDHB, LDHC, LDHD, AGXT, GRHPR, etc and markers for glutathione status, and markers of enzyme cofactors like B vitamins, minerals, and electrolytes.
| ||
|
|
|
|
10 Toxins Lurking in Your Cookware (& How to Make Them Safer)
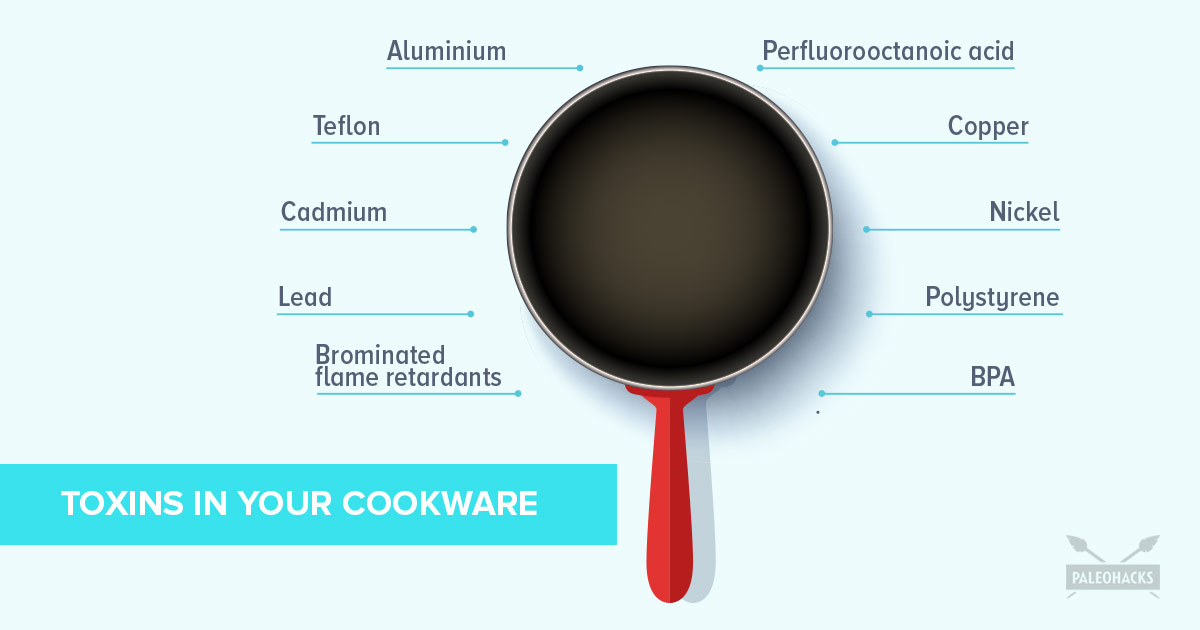
10 Toxins Lurking in Your Cookware (& How to Avoid Them)
Warning: That non-stick pan is leaching dangerous chemicals into your food.
Norseman Leif Eriksson spotted North America 500 years before Columbus — and unlike earlier sailors who had looked at the rocky terrain from the safety of their sailing vessels, he and his crew set foot on Baffin Island, Labrador, and Newfoundland, where they spent the winter.
Those Nordic walks through uncharted territory provided the adventurers with plenty of physical and mental stimulation, as well as tales to tell when they returned home to Greenland.
Special: Doctors Witness Amazing Joint Pain Changes
Nordic walking may be uncharted territory for you too. But fortunately, it's just as rewarding physically and mentally today, with none of the risks Eriksson faced.
If you want to increase the benefits of your 10,000 steps a day, get ahold of two Nordic walking poles. Holding the poles behind you as an extension of your arms, move them to match your foot stride. With every step you apply force to the pole, propelling yourself forward and increasing workout intensity, with no added strain on your joints in your lower body.
You will:
• Activate 90% of your body's muscles and burn 46% more calories than regular walking
• Increase your aerobic activity by 25%
• Tone your arms, shoulders, and back muscles while you improve your core strength and posture
For people with Parkinson's disease, Nordic walking has been found to help with posture and balance, quality of gait, and quality of life.
So, explore the benefits of Nordic walking as you stride out through your favorite park or discover new territory for your physical activity.
Those Nordic walks through uncharted territory provided the adventurers with plenty of physical and mental stimulation, as well as tales to tell when they returned home to Greenland.
Special: Doctors Witness Amazing Joint Pain Changes
Nordic walking may be uncharted territory for you too. But fortunately, it's just as rewarding physically and mentally today, with none of the risks Eriksson faced.
If you want to increase the benefits of your 10,000 steps a day, get ahold of two Nordic walking poles. Holding the poles behind you as an extension of your arms, move them to match your foot stride. With every step you apply force to the pole, propelling yourself forward and increasing workout intensity, with no added strain on your joints in your lower body.
You will:
• Activate 90% of your body's muscles and burn 46% more calories than regular walking
• Increase your aerobic activity by 25%
• Tone your arms, shoulders, and back muscles while you improve your core strength and posture
For people with Parkinson's disease, Nordic walking has been found to help with posture and balance, quality of gait, and quality of life.
So, explore the benefits of Nordic walking as you stride out through your favorite park or discover new territory for your physical activity.
| Hey Jimi, We’re finally at the end of our groundbreaking 9-episode docuseries, BRAVE: Live Courageously, Heal Miraculously… and I want to say a massive “Thank YOU” for your incredible support. We had hundreds of thousands of viewers coming back, episode after episode, and the feedback we’ve received so far is truly humbling. But we’re not done yet… Because we saved the best episode for last. In today’s Episode #9 - BRAVE AWARENESS: Exposing the Global Governments’ Coercive Agendas… our experts dive deep into the next chain of events the globalists are primed to trigger. These evil population-control fanatics are more anxious than ever to “cull” us in the billions since C0V!D didn’t work as well as they hoped… Although the “vaccine” will continue to cause “sudden” “unexplained” “natural” deaths for years to come… But we’re right on their heels to prevent that. Click the link below to watch the shocking revelations in Episode 9 now: |
FDA refuses to release autopsy reports that could shed light on COVID vaccine victims
FDA refuses to release autopsy reports that could shed light on COVID vaccine victims - LifeSite
The FDA cited privacy concerns, ignoring the option of redacting personal information.
| ||
|
| ||
|
Hi Jimi,
Have you been sick for years with the weirdest mystery symptoms no one can figure out?
It takes an average of 10 years to get diagnosed with a condition known as MCAS. One of the most common symptoms is brain fog.
Do you feel like you're walking around in a constant fog? Like it takes all your energy to focus on work throughout the day, and you have nothing left when you get home?
Brain fog is one of the most debilitating and frustrating things to deal with because it impacts every part of your life.
But there’s hope!
Your chronic fatigue and/or brain fog may be from Mast Cell Activation Syndrome – a disorder that affects up to 17% of the population!
That was my friend Beth O’Hara's story not long ago.
She designed Mast Cell 360 to be the kind of practice she wished had existed when she was severely ill with Mast Cell Activation Syndrome, Histamine Intolerance, Mold Toxicity, Neural Inflammation, Lyme, Fibromyalgia, and Chronic Fatigue.
And now, she's hosting a life-saving event called Reversing Mast Cell Activation and Histamine Intolerance Summit.
After you register here, be CERTAIN to listen to her moving and inspiring story right on the next page. <=
The Reversing Mast Cell Activation and Histamine Intolerance Summit brings together more than 40 of the world’s leading experts in MCAS, Histamine Intolerance, chronic illness, and more to help you reverse your symptoms and restore your health.
Get your ticket here at no cost. <=
See you at the summit!
In health,
Have you been sick for years with the weirdest mystery symptoms no one can figure out?
It takes an average of 10 years to get diagnosed with a condition known as MCAS. One of the most common symptoms is brain fog.
Do you feel like you're walking around in a constant fog? Like it takes all your energy to focus on work throughout the day, and you have nothing left when you get home?
Brain fog is one of the most debilitating and frustrating things to deal with because it impacts every part of your life.
But there’s hope!
Your chronic fatigue and/or brain fog may be from Mast Cell Activation Syndrome – a disorder that affects up to 17% of the population!
That was my friend Beth O’Hara's story not long ago.
She designed Mast Cell 360 to be the kind of practice she wished had existed when she was severely ill with Mast Cell Activation Syndrome, Histamine Intolerance, Mold Toxicity, Neural Inflammation, Lyme, Fibromyalgia, and Chronic Fatigue.
And now, she's hosting a life-saving event called Reversing Mast Cell Activation and Histamine Intolerance Summit.
After you register here, be CERTAIN to listen to her moving and inspiring story right on the next page. <=
The Reversing Mast Cell Activation and Histamine Intolerance Summit brings together more than 40 of the world’s leading experts in MCAS, Histamine Intolerance, chronic illness, and more to help you reverse your symptoms and restore your health.
Get your ticket here at no cost. <=
See you at the summit!
In health,
|
|
Does Eating Healthy When Stressed Make You Feel Better?
Eating a healthy diet can reduce the negative effects of stress on the body and builds a solid, more enduring foundation for your longterm health. Eating a well-balanced diet promotes health by reducing oxidation and inflammation, and by helping to reduce weight gain. In this video, I am joined by Evan to talk about how we can better handle stress to keep our health in tact. I can't wait for you to check it out!
>> Click here to check out the podcast! <<
Eating a healthy diet can reduce the negative effects of stress on the body and builds a solid, more enduring foundation for your longterm health. Eating a well-balanced diet promotes health by reducing oxidation and inflammation, and by helping to reduce weight gain. In this video, I am joined by Evan to talk about how we can better handle stress to keep our health in tact. I can't wait for you to check it out!
>> Click here to check out the podcast! <<
8 Powerful Benefits of Maitake Mushroom: The Shroom King

8 Powerful Benefits of Maitake Mushroom: The Shroom King
There are many incredible benefits of maitake mushroom that most people don't know about. Learn all that the "king of mushrooms" can do.
Enough of your mystery symptoms, Jimi! 
It’s time, Jim…
For more than 4 years, I’ve seen many of you in the Healing Rosie community fight to get your life back from debilitating mystery symptoms, taking two steps forward and one step back.
I get messages and see posts in the Healing Rosie Facebook group from so many of you looking for more ideas and support for “where to look next.”
You still:
 Can’t sleep at night and are exhausted all the time
Can’t sleep at night and are exhausted all the time
 Feel like you have nothing left for your family after a day at work
Feel like you have nothing left for your family after a day at work
 Feel unattractive or undesirable because of your skin and the number on the scale
Feel unattractive or undesirable because of your skin and the number on the scale
 Struggle to release stubborn pounds, constantly fighting the yo-yo
Struggle to release stubborn pounds, constantly fighting the yo-yo
 Are exhausted from brain fog, forgetfulness, and an inability to concentrate or focus for longer periods of time
Are exhausted from brain fog, forgetfulness, and an inability to concentrate or focus for longer periods of time
You struggle with menstrual pain or brutal menopause, hair loss and recurring headaches that don’t go away…
The anxiety creeps in, making you feel like your efforts aren’t paying off and you’re wasting precious energy … tempting you to give up, accept your fate and survive your years instead of thriving…
I know how all of this feels Jim.
I spent a decade engaged in the struggle … making progress and then watching it slip away.
No one should have to struggle so hard to get help for healing.
We don’t have to settle for the gaslighting of “you’re normal” and “everything is fine” despite sleeplessness, pain and debilitating symptoms that won’t go away.
I was actually relieved to uncover toxins like heavy metals and mold swimming in my system, causing the stress and inflammation that have been so confounding the last few years, especially.
I’ve learned so much from this roller coaster experience and I’m so committed to sharing everything I can to empower you to decode what’s going on in your body, too!
In October, I am hosting the F*REE 5-Day Radical Healing Detox Challenge to teach you everything I’ve learned to help you have your best sleep ever, wake up energized and finally feel like yourself again!
During this 5-Day Challenge, you’ll learn an actionable framework to help you uncover your stressors and lay the critical foundation you need to detoxify your life. Check out the schedule:
Day 1: The 5 Labs to Run to Uncover the Root Cause of Your Issues
Day 2: The Best Strategies for Powerful Detox
Day 3: Detox Secrets for Getting Rid of Stubborn Stressors
Day 4: Jedi Sleep Hacks to Get Your Best Sleep Ever
Day 5: Turning Down Your Stress Response so Your Body Can Heal
Day 6 BONUS: Detoxify Your Home Without Breaking the Bank
If you want to sleep better, have great energy, a sharp brain, and fit back into your clothes…
Stop the whack-a-mole and hone in on the strategies, tests and lifestyle hacks that are actually going to move the needle and help you start feeling like your best self again…
I invite you to join the F*REE 5-Day Radical Healing Detox Challenge!
Learn from me and other sleep experts live starting Monday, Oct 10th at 12 pm CST / 10 am PST / 11 am MST / 6 pm BST the best strategies for detoxing your stress and stressors.
Click the link below to reserve your spot to join the upcoming 5-Day Better Sleep challenge!
>> I’m joining the 5-Day Radical Healing Detox Challenge!<<
I created this program just for you!
It’s time, Jim…
For more than 4 years, I’ve seen many of you in the Healing Rosie community fight to get your life back from debilitating mystery symptoms, taking two steps forward and one step back.
I get messages and see posts in the Healing Rosie Facebook group from so many of you looking for more ideas and support for “where to look next.”
You still:
You struggle with menstrual pain or brutal menopause, hair loss and recurring headaches that don’t go away…
The anxiety creeps in, making you feel like your efforts aren’t paying off and you’re wasting precious energy … tempting you to give up, accept your fate and survive your years instead of thriving…
I know how all of this feels Jim.
I spent a decade engaged in the struggle … making progress and then watching it slip away.
No one should have to struggle so hard to get help for healing.
We don’t have to settle for the gaslighting of “you’re normal” and “everything is fine” despite sleeplessness, pain and debilitating symptoms that won’t go away.
I was actually relieved to uncover toxins like heavy metals and mold swimming in my system, causing the stress and inflammation that have been so confounding the last few years, especially.
I’ve learned so much from this roller coaster experience and I’m so committed to sharing everything I can to empower you to decode what’s going on in your body, too!
In October, I am hosting the F*REE 5-Day Radical Healing Detox Challenge to teach you everything I’ve learned to help you have your best sleep ever, wake up energized and finally feel like yourself again!
During this 5-Day Challenge, you’ll learn an actionable framework to help you uncover your stressors and lay the critical foundation you need to detoxify your life. Check out the schedule:
Day 1: The 5 Labs to Run to Uncover the Root Cause of Your Issues
Day 2: The Best Strategies for Powerful Detox
Day 3: Detox Secrets for Getting Rid of Stubborn Stressors
Day 4: Jedi Sleep Hacks to Get Your Best Sleep Ever
Day 5: Turning Down Your Stress Response so Your Body Can Heal
Day 6 BONUS: Detoxify Your Home Without Breaking the Bank
If you want to sleep better, have great energy, a sharp brain, and fit back into your clothes…
Stop the whack-a-mole and hone in on the strategies, tests and lifestyle hacks that are actually going to move the needle and help you start feeling like your best self again…
I invite you to join the F*REE 5-Day Radical Healing Detox Challenge!
Learn from me and other sleep experts live starting Monday, Oct 10th at 12 pm CST / 10 am PST / 11 am MST / 6 pm BST the best strategies for detoxing your stress and stressors.
Click the link below to reserve your spot to join the upcoming 5-Day Better Sleep challenge!
>> I’m joining the 5-Day Radical Healing Detox Challenge!<<
I created this program just for you!
Happy Wednesday, my relentless partner in healthy living.
I bet you didn't know this, but October is a special month.
Yes, football is in full swing, baseball playoffs are starting, and basketball will start later this month (can you tell I like sports??).
But, more importantly, October is Spinal Health Awareness Month!
And to celebrate, I'm hosting my next BYOG Party (which stands for "Be Your Own Guarantee" if you missed the reference in the past ).
).
Essentially, it's a get-together I host on Zoom once a month covering a specific healthy living practice.
The entire "party" is designed to increase the likelihood that you live a long pain-free, drug-less, healthy life.
And this month's BYOG Party is no different and is titled STAND TALL: How to Upgrade Your Posture & Spinal Health.
A healthy spine keeps you moving, protects your nervous system, and helps you stand tall, among the other huge benefits for your OVERALL health and life.
So, here's what you'll learn at the BYOG Party (and celebrate your spine...):
I'm hosting the party LIVE on Zoom Wednesday, October 19th at 12pm ET / 9am PT.
I will also record it and send you the REPLAY if you can't attend live.
Plus, there's NO CHARGE to attend or watch the replay.
All you have to do is register here to attend and/or watch the replay.
And, as always, Be Your Own Guarantee for your health, SPINE, and life.
- Dr. Ryan
P.S. Being a practicing chiropractor for 22+ years, I can share this truth with you: the MOST MISUNDERSTOOD healthy living practice is posture and spinal health. I'm here to help you unlock this amazing gateway to OVERALL health. Click here and let me know you're coming to the party.
I bet you didn't know this, but October is a special month.
Yes, football is in full swing, baseball playoffs are starting, and basketball will start later this month (can you tell I like sports??).
But, more importantly, October is Spinal Health Awareness Month!
And to celebrate, I'm hosting my next BYOG Party (which stands for "Be Your Own Guarantee" if you missed the reference in the past
Essentially, it's a get-together I host on Zoom once a month covering a specific healthy living practice.
The entire "party" is designed to increase the likelihood that you live a long pain-free, drug-less, healthy life.
And this month's BYOG Party is no different and is titled STAND TALL: How to Upgrade Your Posture & Spinal Health.
A healthy spine keeps you moving, protects your nervous system, and helps you stand tall, among the other huge benefits for your OVERALL health and life.
So, here's what you'll learn at the BYOG Party (and celebrate your spine...):
- How your spine and posture are DIRECTLY RELATED to health and disease (including chronic pain, heart disease, breathing issues, depression, mood, and decreased lifespan.)
- Why your spinal curves and posture is the UNDERUTILIZED key to active longevity & mobility for life
- Simple solutions to UPGRADE your posture habits
I'm hosting the party LIVE on Zoom Wednesday, October 19th at 12pm ET / 9am PT.
I will also record it and send you the REPLAY if you can't attend live.
Plus, there's NO CHARGE to attend or watch the replay.
All you have to do is register here to attend and/or watch the replay.
And, as always, Be Your Own Guarantee for your health, SPINE, and life.
- Dr. Ryan
P.S. Being a practicing chiropractor for 22+ years, I can share this truth with you: the MOST MISUNDERSTOOD healthy living practice is posture and spinal health. I'm here to help you unlock this amazing gateway to OVERALL health. Click here and let me know you're coming to the party.
Jimi,
All disease begins in the Gut… right?
Or is it the Brain?
Or the Immune system?
Here’s the TRUTH...
For many of us, it may be all three.
And all three--the Brain, Immunity, and the Gut--need to be addressed no matter what your symptoms are.
There is a Brain-Immune-Gut axis that interconnects all 3 systems, and when one part of the system goes down, the other parts suffer as well.
Join me to discover the BIG Idea in healing chronic conditions at The Brain Immune Gut Masterclass, hosted by our friend Dr. Peter Kan just started!!!

This event focuses on speaking with experts on practical strategies that you can use to improve your Brain-Immune-Gut Axis, which will have a far-reaching impact on the whole body.
Also, Dr. Kan will personally teach many different mastery sessions, where you will learn a ton about impactful self-tests, practices, and much more that you can implement right away!
The bottom line is that whether you are trying to heal from a chronic illness or fight an acute infection, you must address the Brain-Immune-Gut dysfunction that is driving it all.
I'll see ya' there!
<<Grab Your Ticket>>
All disease begins in the Gut… right?
Or is it the Brain?
Or the Immune system?
Here’s the TRUTH...
For many of us, it may be all three.
And all three--the Brain, Immunity, and the Gut--need to be addressed no matter what your symptoms are.
There is a Brain-Immune-Gut axis that interconnects all 3 systems, and when one part of the system goes down, the other parts suffer as well.
Join me to discover the BIG Idea in healing chronic conditions at The Brain Immune Gut Masterclass, hosted by our friend Dr. Peter Kan just started!!!
This event focuses on speaking with experts on practical strategies that you can use to improve your Brain-Immune-Gut Axis, which will have a far-reaching impact on the whole body.
Also, Dr. Kan will personally teach many different mastery sessions, where you will learn a ton about impactful self-tests, practices, and much more that you can implement right away!
The bottom line is that whether you are trying to heal from a chronic illness or fight an acute infection, you must address the Brain-Immune-Gut dysfunction that is driving it all.
I'll see ya' there!
<<Grab Your Ticket>>
After researching food for years this is so true
Also not listed here but an apple every day supports healthy liver function and helps to prevent liver problems
An Apple a Day Just Might Keep the Doctor Away
Also not listed here but an apple every day supports healthy liver function and helps to prevent liver problems
We’re going to go over six all-natural things that you can start immediately to boost your immune system.
1. Eat healthy, nourishing foods.
It’s so important that your diet includes plenty of fruits, vegetables, legumes, whole grains, lean protein, and healthy fats.
Incorporating a balance of these vitamin-rich foods in your meals each day can make all the difference when your immune system is trying to fight off infection.
Make a concerted effort to balance your diet by planning out your meals in advance. You’ll make healthier choices and you won’t have to have the dreaded “What should we have for dinner?” talk at 6 pm each night!
2. Drink plenty of water!
We hear it all the time, but it’s critical that we stay hydrated. Our bodies dehydrate through sleeping and even breathing, so we have to make sure to replenish fluids throughout the day.
Dehydration can slow down infection-fighting immune cells, and it’s important to do everything we can to keep those strong!
Try carrying a reusable water bottle with you so that you’ll remember to stay hydrated.
3. Get adequate sleep.
We all lead hectic lives, and it seems like on a busy day, the first thing to suffer is our sleep quality and quantity.
Sleep allows our body to heal and even create infection-fighting cells. Make sleep a priority, even when you have a never-ending to-do list.
Consider setting a “bedtime” for yourself this week that’s 30-45 minutes earlier than usual and see how you feel.
4. Try to relax.
As humans, we don’t enjoy feeling stressed and we certainly don’t strive to live stressful lives, but still, almost 80% of Americans feel stressed at least one day per week.
Your immune system response weakens with increased levels of stress. It’s essential that you try to reduce stress in your life, as difficult as that may seem.
Consider meditation or yoga to make stress relief a part of your daily routine.
5. Move your body frequently.
Exercise is key! Not only will you gain strength and endurance, but you’ll feel better, too!
Sweating allows your body to release toxins that could be making you feel chronically sick, and increasing circulation allows for the immune system cells to move more easily throughout your body.
If you don’t have time for daily trips to the gym, try something more manageable like an at-home workout or a walk after dinner with the family.
6. Stay consistent!
Although you can employ all of these habits today to boost your immune system, it’s important to keep up with them consistently.
Implementing each of these for a day or even a week will help your body heal and fight sickness and infection, but consistency will make all the difference in living a long life full of vitality and wellness.
1. Eat healthy, nourishing foods.
It’s so important that your diet includes plenty of fruits, vegetables, legumes, whole grains, lean protein, and healthy fats.
Incorporating a balance of these vitamin-rich foods in your meals each day can make all the difference when your immune system is trying to fight off infection.
Make a concerted effort to balance your diet by planning out your meals in advance. You’ll make healthier choices and you won’t have to have the dreaded “What should we have for dinner?” talk at 6 pm each night!
2. Drink plenty of water!
We hear it all the time, but it’s critical that we stay hydrated. Our bodies dehydrate through sleeping and even breathing, so we have to make sure to replenish fluids throughout the day.
Dehydration can slow down infection-fighting immune cells, and it’s important to do everything we can to keep those strong!
Try carrying a reusable water bottle with you so that you’ll remember to stay hydrated.
3. Get adequate sleep.
We all lead hectic lives, and it seems like on a busy day, the first thing to suffer is our sleep quality and quantity.
Sleep allows our body to heal and even create infection-fighting cells. Make sleep a priority, even when you have a never-ending to-do list.
Consider setting a “bedtime” for yourself this week that’s 30-45 minutes earlier than usual and see how you feel.
4. Try to relax.
As humans, we don’t enjoy feeling stressed and we certainly don’t strive to live stressful lives, but still, almost 80% of Americans feel stressed at least one day per week.
Your immune system response weakens with increased levels of stress. It’s essential that you try to reduce stress in your life, as difficult as that may seem.
Consider meditation or yoga to make stress relief a part of your daily routine.
5. Move your body frequently.
Exercise is key! Not only will you gain strength and endurance, but you’ll feel better, too!
Sweating allows your body to release toxins that could be making you feel chronically sick, and increasing circulation allows for the immune system cells to move more easily throughout your body.
If you don’t have time for daily trips to the gym, try something more manageable like an at-home workout or a walk after dinner with the family.
6. Stay consistent!
Although you can employ all of these habits today to boost your immune system, it’s important to keep up with them consistently.
Implementing each of these for a day or even a week will help your body heal and fight sickness and infection, but consistency will make all the difference in living a long life full of vitality and wellness.
|
| Hi Jimi, |
| Why is vitamin A important? How much do you need? Which types are best? What are your best sources? And should you take a supplement? |
| Get the whole vitamin A story here. |
| Yours for nutritional wisdom, |
| Ocean Robbins |
| P.S. It’s widely believed that vegans are at risk for vitamin A deficiency. But are they? Click here to find out what you need to know about vitamin A. |